Science Notes
PERIODIC CLASSIFICATION OF ELEMENTS Notes 9th Science Lesson 10 Notes in English
PERIODIC CLASSIFICATION OF ELEMENTS
Introduction
- We live in the world of substances with great diversity. Substances are formed by the combination of various elements.
- All the elements are unique in their nature and property.
- To categorize these elements according to their properties, scientists started to look for a way.
- In 1800, there were only 31 known elements.
- By 1865, their number became 63.
- Now 118 elements have been discovered.
- As different elements were being discovered, scientists gathered more and more information about the properties of these elements.
- They found it difficult to organize all that was known about the elements.
- They started looking for some pattern in their properties, on the basis of which they could study such a large number of elements with ease.
- Let us discuss the concepts of classification of elements proposed by various scientists from early to modern period.
Early Concepts of Classification of Elements
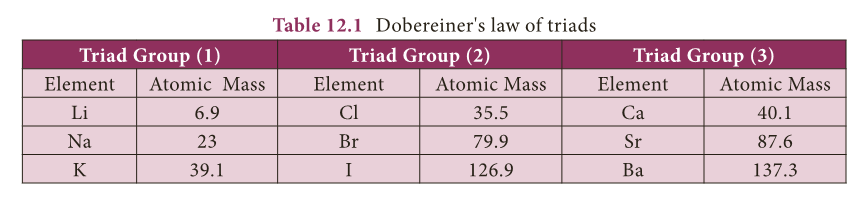
Dobereiner’s Triads
- In 1817, Johann Wolfgang Dobereiner, a German chemist, suggested a method of grouping elements based on their relative atomic masses.
- He arranged the elements into groups containing three elements each. He called these groups as ‘triads’ (tri – three).
- Dobereiner showed that when the three elements in a triad are arranged in the ascending order of their atomic masses, the atomic mass of the middle element is nearly the same as average of atomic masses of other two elements.
- This statement is called the Dobereiner’s law of triads. Table 12.1 shows the law of triads proposed by Dobereiner.
- Example: In the triad group (1), arithmetic mean of atomic masses of 1st and 3rd elements, (6.9 + 39.1)/2 = 23.
- So the atomic mass of Na (middle element) is 23.
Limitations:
- Dobereiner could identify only three triads from the elements known at that time and all elements could not be classified in the form of triads.
- The law was not applicable to elements having very low and very high atomic mass.
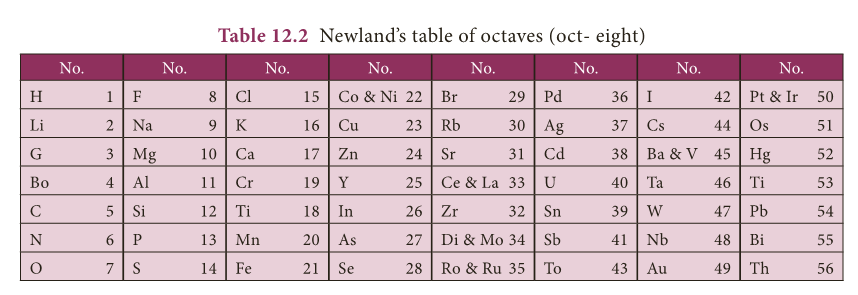
Newlands’ Law of Octaves
- In 1866, John Newlands arranged 56 known elements in the increasing order of their atomic mass.
- He observed that every eighth element had properties similar to those of the first element like the eighth note in an octave of music is similar to the first.
- This arrangement was known as ‘law of octaves’. The octave of Indian music system is sa, re, ga, ma, pa, da, ni, sa.
- The first and last notes of this octave are same i.e. sa.
- Likewise, in the Newlands’ table of octaves, the element ‘F’ is eighth from the element ‘H’, thus they have similar properties.
Limitations:
- There are instances of two elements being fitted into the same slot, e.g. cobalt and nickel.
- Some elements, totally dissimilar in their properties, were fitted into the same group. (Arrangement of Co, Ni, Pd, Pt and Ir in the row of halogens)
- The law of octaves was not valid for elements that had atomic masses higher than that of calcium.
- Newlands’ table was restricted to only 56 elements and did not leave any room for new elements.
- Discovery of inert gases (Neon. Argon….) at later stage made the 9th element similar to the first one. Eg: Neon between Fluorine and Sodium.

Mendeleev’s Periodic Table
- In 1869, Russian chemist, Dmitri Mendeleev observed that the elements of similar properties repeat at regular intervals when the elements are arranged in the order of their atomic masses.
- Based on this, he proposed the law of periodicity which states that “the physical and chemical properties of the elements are the periodic functions of their atomic masses”.
- He arranged 56 elements known at that time according to his law of periodicity.
- This was best known as the short form of periodic table.
(a) Features of Mendeleev’s Periodic Table:
- It has eight vertical columns called ‘groups’ and seven horizontal rows called ‘period’.
- Each group has two subgroups ‘A’ and ‘B’.
- All the elements appearing in a group were found to have similar properties.
- For the first time, elements were comprehensively classified in such a way that elements of similar properties were placed in the same group.
- It was noticed that certain elements could not be placed in their proper groups in this manner.
- The reason for this was wrongly determined atomic masses.
- Consequently those wrong atomic masses were corrected. Eg: The atomic mass of beryllium was known to be 14.
- Mendeleev reassessed it as 9 and assigned beryllium a proper place.
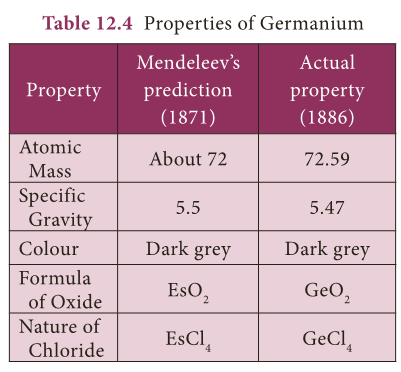
- Columns were left vacant for elements which were not known at that time and their properties also were predicted.
- This gave motivation to experiment in Chemistry. Eg: Mendeleev gave names Eka Aluminium and Eka Silicon to those elements which were to be placed below
- Aluminium and Silicon respectively in the periodic table and predicted their properties.
- The discovery of Germanium later on, during his life time, proved him correct.
(b) Limitations:
- Elements with large difference in properties were included in the same group. Eg: Hard metals like copper (Cu) and silver (Ag) were included along with soft metals like sodium (Na) and potassium (K).
- No proper position could be given to the element hydrogen.
- Non-metallic hydrogen was placed along with metals like lithium (Li), sodium (Na) and potassium (K).
- The increasing order of atomic mass was not strictly followed throughout. Eg. Co & Ni, Te & I.
- No place for isotopes in the periodic table.
Modern Periodic Table
- In 1913, the English Physicist Henry Moseley, through his X-ray diffraction experiments, proved that the properties of elements depend on the atomic number and not on the atomic mass.
- Consequently, the modern periodic table was prepared by arranging elements in the increasing order of their atomic number.
- This modern periodic table is the extension of the original Mendeleev’s periodic table and known as the long form of periodic table.
Modern Periodic Law
- Atomic number of an element (Z) indicates the number of protons (positive charge) or the number of electrons (negative charge).
- The physical and chemical properties of elements depend not only on the number of protons but also on the number of electrons and their arrangements (electronic configuration) in atoms.
- Hence, the modern periodic law can be stated as follows: “The chemical and physical properties of the elements are the periodic functions of their atomic numbers”.
- Based on the modern periodic law, the modern periodic table is derived.
Features of Modern Periodic Table
- All the elements are arranged in the increasing order of their atomic number.
- The horizontal rows are called periods.
- There are seven periods in the periodic table.
- The elements are placed in periods based on the number of shells in their atoms.
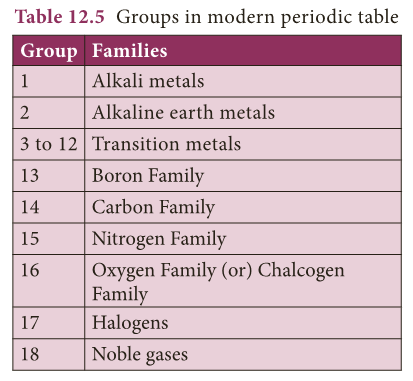
- Vertical columns in the periodic table starting from top to bottom are called groups.
- There are 18 groups in the periodic table.
- Based on the physical and chemical properties of elements, they are grouped into various families.


Classification of elements into blocks
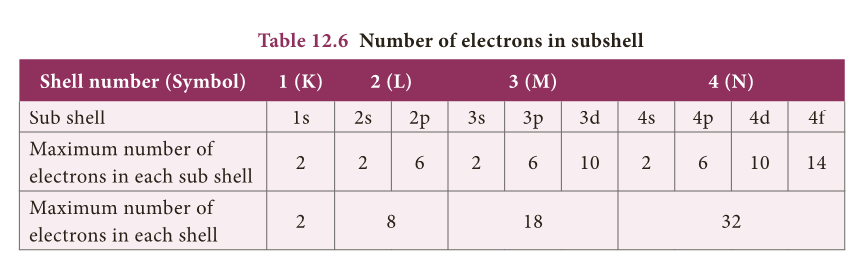
- We know that the electrons in an atom are accommodated in shells around the nucleus.
- Each shell consists of one or more subshells in which the electrons are distributed in certain manner.
- These subshells are designated as s, p, d, and f.
- Based on the arrangement of electrons in subshells, the elements of periodic table are classified into four blocks namely s, p, d and f blocks.
(1) s-Block Elements:
- It includes group 1 (alkali metals) and group 2 (alkaline earth metals) elements.
- They are also called as representative elements.
- The elements of group 1 (except hydrogen) are metals.
- They react with water to form solutions that change the colour of a litmus paper from red to blue.
- These solutions are said to be highly alkaline or basic. Hence they are called alkali metals.
- The elements of group 2 are also metals.
- They combine with oxygen to form oxides, formerly called ‘earths’, and these oxides produce alkaline solutions when they are dissolved in water.
- Hence, these elements are called alkaline earth metals.
(2) p-Block Elements:
- These elements are in group 13 to 18 in the periodic table.
- They include boron, carbon, nitrogen, oxygen, fluorine families in addition to noble gases (Except helium).
- They are also called as representative elements.
- The p-block is home to the biggest variety of elements and is the only block that contains all three types of elements: metals, nonmetals, and metalloids.
(3) d-Block Elements:
- It includes group 3 to group 12 elements.
- They are found in the centre of the periodic table.
- Their properties are intermediate to that of s block and p block elements and so they are called transition elements.
(4) f – Block Elements:
- It includes 14 elements after (Lanthanum) La (57), called Lanthanoides and 14 elements after (Actinium) Ac (89), called Actinoides.
- They are placed at the bottom of the periodic table.
- They are also called as inner Transition elements.
Advantages of the Modern Periodic Table
- The table is based on a more fundamental property i.e., atomic number.
- It correlates the position of the element with its electronic configuration more clearly.
- The completion of each period is more logical.
- In a period, as the atomic number increases, the energy shells are gradually filled up until an inert gas configuration is reached.
- It is easy to remember and reproduce.
- Each group is an independent group and the idea of subgroups has been discarded.
- One position for all isotopes of an element is justified, since the isotopes have the same atomic number.
- The position of the eighth group (in Mendeleev‘s table) is also justified in this table.
- All transition elements have been brought in the middle as the properties of transition elements are intermediate between left portion and right portion elements of the periodic table.
- The table completely separates metals from nonmetals.
- The nonmetals are present in upper right corners of the periodic table.
- The positions of certain elements which were earlier misfit (interchanged) in the Mendeleev’s periodic table are now justified because it is based on atomic number of the elements.
- Justification has been offered for placing lanthanides and actinides at the bottom of the periodic table.
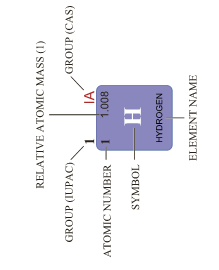
Position of hydrogen in the periodic table
- Hydrogen is the lightest, smallest and first element of the periodic table.
- Its electronic configuration (1s1) is the simplest of all the elements.
- It occupies a unique position in the periodic table.
- It behaves like alkali metals as well as halogens in its properties.
- In the periodic table, it is placed at the top of the alkali metals.
- Hydrogen can lose its only one electron to form a hydrogen ion (H+) like alkali metals.
- It can also gain one electron to form the hydride ion (H-) like halogens.
- Alkali metals are solids while hydrogen is a gas.
- Hence the position of hydrogen in the modern periodic table is still under debate as the properties of hydrogen are unique.
Position of Rare Gases
- The elements Helium, Neon, Argon, Krypton, Xenon and Radon of group 18 in the periodic table are called as Noble gases or Rare gases.
- They are monoatomic gases and do not react with other substances easily, due to completely filled subshells.
- Hence they are called as inert gases.
- They are found in very small quantities and hence they are called as rare gases.
Metals, Non-Metals and Metalloids
Metals
- Metals are typically hard, shiny, malleable (can be made as sheet), fusible and ductile (can be drawn into wire) with good electrical and thermal conductivity.
- Except mercury, most of the metals are solids at room temperature.
- Metals occupy larger area in the periodic table and are categorized as:
- Alkali metals. e.g. Lithium to Francium (top to bottom)
- Alkaline earth metals. e.g: Beryllium to Radium (top to bottom)
- Transition Metals. Group 3 to 12 (iv) p-block metals. e.g: Al, Ga, In, Tl, Sn, Pb and Bi.
Non-metals
- A non-metal is an element that does not have the characters like hardness, shiny, malleable, suitable and ductile.
- In other words, a non-metal is an element that does not have the properties of metal.
- All non metals are arranged in p-block only. E.g. C, N, O, P, S, Se, Halogen (F, Cl, Br and I) and inert gases (He to Rn).
Metalloids
- Elements which have the properties of both metals and non-metals are called as metalloids. (eg) Boron, Arsenic.
Alloys
- During 3500 BC(BCE), people used an alloy named ‘bronze’.
- The idea of making an alloy was quite old. The majority of the metallic substances used today are alloys.
- Alloys are mixtures of two or more metals and are formed by mixing molten metals thoroughly.
- Rarely nonmetals are also mixed with metals to produce alloys.
- It is generally found that alloying produces a metallic substance that has more useful properties than the original pure metals from which it is made.
- For example, the alloy brass is made from copper and zinc.
Advantages of alloys
- Alloys do not get corroded or get corroded to very less extent.
- They are harder and stronger than pure metals (Example: Gold is mixed with copper and it is harder than pure gold).
- They have less conductance than pure metals (Example: Copper is good conductor of heat and electricity where as brass and bronze are not good conductors).
- Some alloys have lower melting point than pure metals (Example: Solder is an alloy of lead and tin which has lower melting point than each of the metals).
- When metal is alloyed with mercury, it is called amalgam.
EXTRA POINTS:
- Dobereiner’s Law of Triads: The atomic mass of the middle element is nearly the same as average of atomic masses of other two elements.
- Newlands’ Law of Octaves: Every eighth element had properties similar to those of the first element like the eighth note in an octave of music is similar to the first.
- Mendeleev’s Law of Periodicity: The physical and chemical properties of the elements are the periodic functions of their atomic masses.
- Modern Periodic Law: The chemical and physical properties of the elements are the periodic functions of their atomic numbers.
- Periods: Horizontal rows in the modern periodic table.
- Columns: Vertical columns in the modern periodic table
- s block elements: Elements whose valence electrons are added to s subshell.
- p block elements: Elements whose valence electrons are filled in p subshells.
- d block elements: Elements having their valence electrons in the d subshells.
- f block elements: Elements having their valence electrons in the f subshells.