Science Notes
Heat Notes 9th Science Lesson 7 Notes in English
Heat Notes 9th Science Lesson 7 Notes in English
Introduction
- All substances in our surrounding are made up of molecules.
- These molecules are generally at motion and posses kinetic energy.
- At the same time each molecule exerts a force of attraction on other molecules and so they posses potential energy.
- The sum of the kinetic and potential energy is called the internal energy of the molecules.
- This internal energy, when flows out, is called heat energy.
- This energy is more in hot substances and less in cold substances and flows from hot substances to cold substances.
- In this lesson you will study about how this heat transfer takes place.
- Also you will study about the effect of heat, heat capacity, change of state and latent heat.
Effects of Heat
- When a substance is heated, the following things can happen.
Expansion:
- When heat is added to a substance, the molecules gain energy and vibrate and force other molecules apart.
- As a result, expansion takes place. You would have seen some space being left in railway tracks.
- It is because, during summer time, more heat causes expansion in tracks.
- Expansion is greater for liquids than solids and it is maximum in gases.

Change in State:
- When you heat ice cubes, they become water and water on further heating changes into vapour.
- So, solid becomes liquid and liquid becomes gas, when heat is added.
- The reverse takes place when heat is removed.
Change in Temperature:
- When heat energy is added to a substance, the kinetic energy of its particles increases and so the particles move at higher speed.
- This causes rise in temperature.
- When a substance is cooled, that is, when heat is removed, the molecules lose heat and its temperature falls.
Chemical changes:
- Since heat is a form of energy it plays a major role in chemical changes.
- In some cases, chemical reactions need heat to begin and also heat determines the speed at which reactions occur.
- When we cook food, we light the wood and it catches fire and the food particles become soft because of the heat energy.
- These are all the chemical changes taking place due to heat.
Transfer of Heat
Activity 1
- Take a glass of water and put some ice cubes into it.
- Observe it for some time. What happens?
- The ice cubes melt and disappear.
- Why did it happen? It is because heat energy in the water is transferred to the ice.
- Heat does not stay where we put it.
- Hot things get colder and cold things get hotter.
- Heat is transferred from one place to another till their temperatures become equal.
- Heat transfer takes place when heat energy flows from an object with higher temperature to an object with lower temperature (Fig. 7.2).
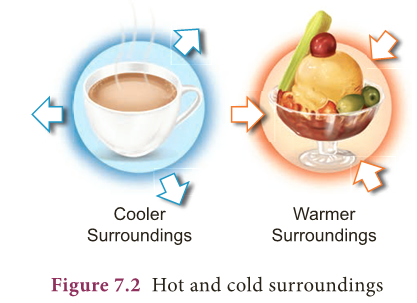
- Heat transfer takes place in three ways:
- Conduction,
- Convection,
- Radiation
Conduction
- In solids, molecules are closely arranged so that they cannot move freely.
- When one end of the solid is heated, molecules at that end absorb heat energy and vibrate fast at their own positions.
- These molecules in turn collide with the neighbouring molecules and make them vibrate faster and so energy is transferred.
- This process continues till all the molecules receive the heat energy.
- The process of transfer of heat in solids from a region of higher temperature to a region of lower temperature without the actual movement of molecules is called conduction.
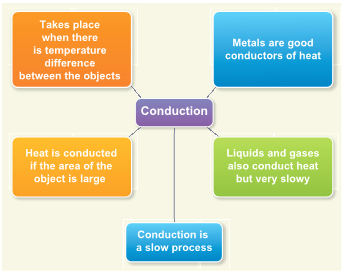
Conduction in daily life
- Metals are good conductors of heat. So, aluminium is used for making utensils to cook food quickly.
- Mercury is used in thermometers because, it is a good conductor of heat.
- We wear woolen clothes in winter to keep ourselves warm. Air, which is a bad conductor, does not allow our body heat to escape.
Activity 2
- Take metal rods of copper, aluminium, brass and iron.
- Fix a match stick to one end of each rod using a little melted wax.
- When the temperature of the far ends reach the melting point of wax, the matches drop off.
- It is observed that the match stick on the copper rod would fall first, showing copper as the best conductor followed by aluminum, brass and iron.
Convection
Activity 3
- Drop a few crystals of potassium permanganate down to the bottom of a beaker containing water.
- When the beaker is heated just below the crystals, by a small flame, purple streaks of water rise upwards and fan outwards.
- In this activity, water molecules at the bottom of the beaker receive heat energy and move upward and replace the molecules at the top.
- Same thing happens in air also. When air is heated, the air molecules gain heat energy allowing them to move further apart.
- Warm air being less dense than cold air will rise.
- Cooler air moves down to replace the air that has risen. It heats up, rises and is again replaced by cooler air, creating a circular flow.
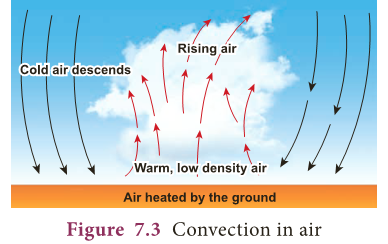
- Convection is the flow of heat through a fluid from places of higher temperature to places of lower temperature by movement of the fluid itself.
Convection in daily life
Hot air balloons:

- Air molecules at the bottom of the balloon get heated by a heat source and rise.
- As the warm air rises, cold air is pushed downward and it is also heated.
- When the hot air is trapped inside the balloon, it rises.
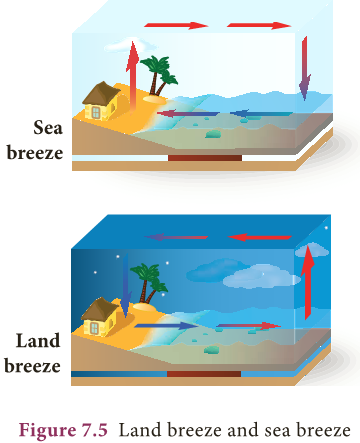
Breezes:
- During day time, the air in contact with the land becomes hot and rises.
- Now the cool air over the surface of the sea replaces it.
- It is called sea breeze.
- During night time, air above the sea is warmer.
- As the warmer air over the surface of the sea rises, cooler air above the land moves towards the sea. It is called land breeze.
Winds:
- Air flows from area of high pressure to area of low pressure.
- The warm air molecules over hot surface rise and create low pressure.
- So, cooler air with high pressure flows towards low pressure area.
- This causes wind flow.
Chimneys:
- Tall chimneys are kept in kitchen and industrial furnaces.
- As the hot gases and smoke are lighter, they rise up in the atmosphere.
Radiation
- Radiation is a method of heat transfer that does not require particles to carry the heat energy.
- In this method, heat is transferred in the form of waves from hot objects in all direction.
- Radiation can occur even in vacuum whereas conduction and convection need matter to be present.
- Radiation consists of electromagnetic waves travelling at the speed of light.
- Thus, radiation is the flow of heat from one place to another by means of electromagnetic waves.
- Transfer of heat energy from the sun reaches us in the form of radiation.
- Radiation is emitted by all bodies above 0 K.
- Some objects absorb radiation and some other objects reflect them.
Radiation in daily life
- White or light coloured cloths are good reflectors of heat. They keep us cool during summer.
- Base of cooking utensils is blackened because black surface absorbs more heat from the surrounding.
- Surface of airplane is highly polished because it helps to reflect most of the heat radiation from the sun.
Concept of Temperature
- Temperature is the degree of hotness or coolness of a body.
- Hotter the body, higher is its temperature.
Unit of Temperature
- The SI unit of temperature is kelvin (K).
- For day to day applications, Celsius (°C) is used.
- Temperature is measured with a thermometer.
Temperature scales
- There are three scales of temperature.
- Fahrenheit scale
- Celsius or Centigrade scale
- Kelvin or Absolute scale
Fahrenheit scale
- In Fahrenheit scale, 32 °F and 212 °F are the freezing point and boiling point respectively. Interval has been divided into 180 parts.
Celsius temperature scale
- In Celsius scale, also called centigrade scale, 0°C and 100 °C are the freezing point and boiling point respectively. Interval has been divided into 100 parts.
- The formula to convert a Celsius scale to Fahrenheit scale is: F=9/ 5 C+32
- The formula for converting a Fahrenheit scale to Celsius scale is: C=5/ 9 (F-32)
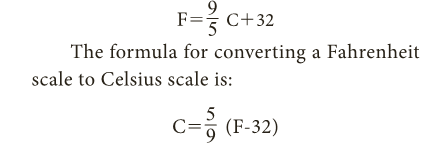
Kelvin scale (Absolute scale)
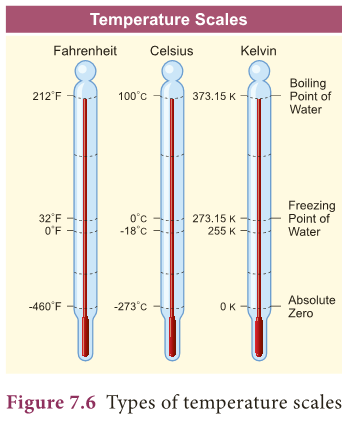
- Kelvin scale is known as the absolute scale.
- On the Kelvin scale 0 K represents absolute zero, the temperature at which the molecules of a substance have their lowest possible energy.
- The solid, liquid, gaseous phases of water can coexist in equilibrium at 273.16 K. Kelvin is defined as 1/273.16 of the triple point temperature.
- The formula for converting a Celsius scale to Kelvin scale is: K = C + 273.15
Absolute zero
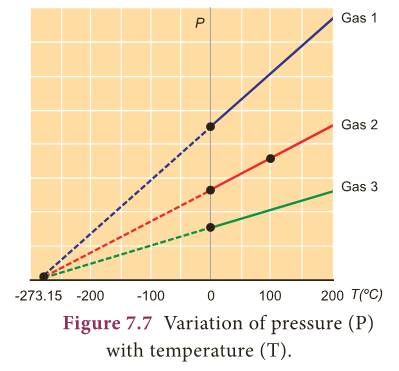
- The temperature at which the pressure and volume of a gas theoretically reaches zero is called absolute zero. This is shown in Figure 7.7.
- For all gases, the pressure extrapolates to zero at the temperature –273.15 °C.
- It is known as absolute zero or 0 K.
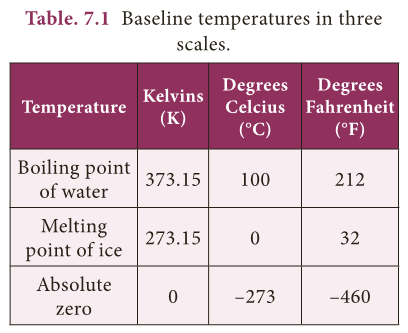
- Some base line temperatures in the three temperature scales are shown in Table 7.1.


Specific Heat Capacity
- You might have felt that the land is cool in the morning and hot during day time.
- But, water in a lake will be almost at a particular temperature both in the morning as well as in the afternoon.
- Both are subjected to same amount of heat energy from the Sun, but they react differently.
- It is because both of them have different properties.
- In general, the amount of heat energy absorbed or lost by a body is determined by three factors.
- Mass of the body
- Change in temperature of the body
- Nature of the material of the body
- We can understand this from the following observations.
Observation: 1
- Quantity of heat required to raise the temperature of 1 litre of water will be more than the heat required to raise the temperature of 500 ml of water.
- If Q is the quantity of heat absorbed and m is the mass of the body, then Q α m (7.1)
Observation: 2
- Quantity of heat energy (Q) required to raise the temperature of 250 ml of water to 100°C is more than the heat energy required to raise the temperature to 50 °C.
- Here, Q α ΔT, where ΔT is the change in temperature of the body.
- Thus, heat lost or gained by a substance when its temperature changes by ΔT is,
- Q α mΔT
- Q = mCΔT
- From the above equations, the absolute temperature and energy of a system are proportional to each other.
- The proportionality constant is the specific heat capacity (C) of the substance.
- ∴ C = Q/mΔT
- Thus, specific heat capacity of a substance is defined as the amount of heat required to raise the temperature of 1 kg of the substance by 1degC or 1 K.
- The SI unit of specific heat capacity is Jkg-1 K-1.
- The most commonly used units of specific heat capacity are J/kg°C and J/g°C.
- Among all the substances, water has the highest specific heat capacity and its value is 4200 J/kg°K.

- So, water absorbs a large amount of heat for unit rise in temperature.
- Thus, water is used as a coolant in car radiators and factories to keep engines and other machinery parts cool.
- It is because of this same reason, temperature of water in the lake does not change much during day time.

Heat capacity or Thermal capacity
- Now, you are familiar with specific heat capacity.
- It is the heat required to raise the temperature of a unit mass of a body by 1°C.
- But, heat capacity is the heat required to raise the temperature of the entire mass of the body by 1°C.
- Thus, heat capacity or thermal capacity is defined as the amount of heat energy required to raise the temperature of a body by 1°C. It is denoted by C’.
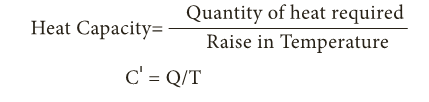
- SI unit of heat capacity is J/K. It is also expressed in cal/oC, kcal/oC or J/oC.

Change of state
- The process of changing of a substance from one physical state to another at a definite temperature is known as change of state.
- For example, water molecules are in liquid state at normal temperature.
- When water is heated to 100°C, it becomes steam which is a gaseous state of matter.
- On reducing the temperature of the steam it becomes water again.
- If we reduce the temperature further to 0 °C, it becomes ice which is a solid state of water.
- Ice on heating, becomes water again. Thus, water changes its state when there is a change in temperature.
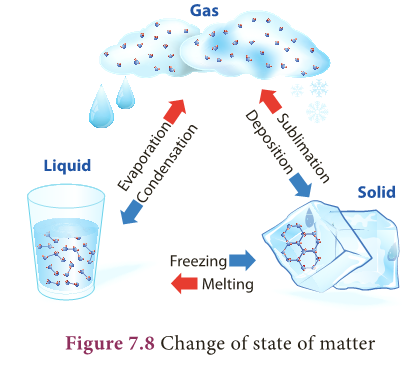
- There are different such processes in the change of state in matter. Figure 7.8 shows various processes of change of state.
Melting – Freezing
- The process in which a solid is converted to liquid by absorbing heat is called melting or fusion.
- The temperature at which a solid changes its state to liquid is called melting point.
- The reverse of melting is freezing.
- The process in which a liquid is converted to solid by releasing heat is called freezing.
- The temperature at which a liquid changes its state to solid is called freezing point.
- In the case of water, melting and boiling occur at 0°C.
Boiling-Condensation
- The process in which a liquid is converted to vapour by absorbing heat is called boiling or vaporization.
- The temperature at which a liquid changes its state to gas is called boiling point.
- The process in which a vapour is converted to liquid by releasing heat is called condensation.
- The temperature at which vapour changes its state to liquid is called condensation point.
- Boiling point as well as condensation point of water is 100°C.
Sublimation
- Some solids like dry ice, iodine, frozen carbon dioxide and naphthalene balls change directly from solid state to gaseous state without becoming liquid.
- The process in which a solid is converted to gaseous state is called sublimation.
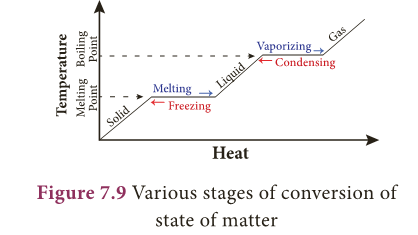
- Various stages of conversion of state of matter by heat with the corresponding change in temperature is shown in Figure 7.9
Latent heat
- The word, ‘latent’ means hidden.
- So, latent heat means hidden heat or hidden energy.
- In order to understand latent heat, let us do the activity given below.
Activity 4
- Take some crushed ice cubes in a beaker and note down the temperature using thermometer.
- It will be 0 deg C. Now heat the ice in the beaker.
- You can observe that ice is melting to form water.
- Record the temperature at regular intervals and it will remain at 0 deg C until whole ice is converted to liquid.
- Now heat the beaker again and record the temperature.
- You can notice that the temperature will rise up to 100 deg C and it will retain the same even after continuous heating until the whole mass of water in the beaker is vaporized.
- In the above activity, temperature is constant at 0°C until entire ice is converted into liquid and again constant at 100°C until all the ice is converted into vapour.
- Why? It is because, when a substance changes from one state to another, a considerable amount of heat energy is absorbed or liberated. This energy is called latent heat.
- Thus, latent heat is the amount of heat energy absorbed or released by a substance during a change in its physical states without any change in its temperature.
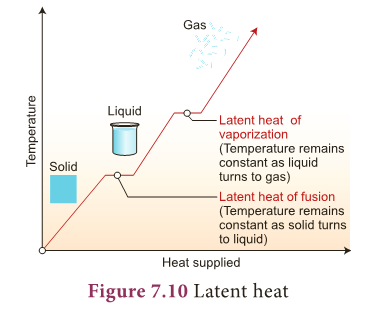
- Heat energy is absorbed by the solid during melting and an equal amount of heat energy is liberated by the liquid during freezing, without any temperature change.
- It is called latent heat of fusion.
- In the same manner, heat energy is absorbed by a liquid during vaporization and an equal amount of heat energy is liberated by the vapour during condensation, without any temperature changes. This is called latent heat of vaporization.
Specific latent heat
- Latent heat, when expressed per unit mass of a substance, is called specific latent heat. It is denoted by the symbol L.
- If Q is the amount of heat energy absorbed or liberated by ‘m’ mass of a substance during its change of phase at a constant temperature, then specific latent heat is given as L = Q/m.
- Thus, specific latent heat is the amount of heat energy absorbed or liberated by unit mass of a substance during change of state without causing any change in temperature.
- The SI unit of specific latent heat is J/kg.


MORE TO KNOW:
DOG
- When a dog keeps out its tongue and breathes hard, the moisture on the tongue turns into water and it evaporates.
- Since, heat energy is needed to turn a liquid into a gas, heat is removed from dog’s tongue. This helps to cool the body of the dog.
Conduction and Convection
- While firing wood, we can observe all the three ways of heat transfer.
- Heat in one end of the wood will be transferred to other end due to conduction.
- The air near the wood will become warm and replace the air above.
- This is convection. Our hands will be warm because heat reaches us in the form of radiation.
Water
- Water in its various form, has different specific heat capacities.
- Water (Liquid state) = 4200 JKg–1 K–1
- Ice (Solid state) = 2100 JKg–1 K–1
- Steam (Gaseous state) = 460 JKg–1 K–1
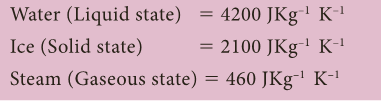
EXTRA POINTS:
- Conduction: Process of transfer of heat in solids from a region of higher temperature to a region of lower temperature without the actual movement of molecules.
- Convection: Flow of heat through a fluid from places of higher temperature to places of lower temperature by movement of the fluid itself.
- Radiation: Flow of heat from one place to another by means of electromagnetic waves.
- Temperature: It is the degree of hotness or coolness of a body.
- Specific heat capacity: The amount of heat required to raise the temperature of 1 kg of the substance by 1°C or 1 K.
- Heat capacity: The amount of heat energy required to raise the temperature of a body by 1°C.
- Melting or fusion: Process in which a solid is converted to liquid by absorbing heat.
- Freezing: Process in which a liquid is converted to solid by releasing heat.
- Vaporization: Process in which a liquid is converted to vapour by absorbing heat.
- Condensation: Process in which a vapour is converted to liquid by releasing heat.
- Latent heat: Amount of heat energy absorbed or released by a substance during a change in its physical state without any change in its temperature.
- Specific latent heat: Amount of heat energy absorbed or liberated by unit mass of substance during change of state without causing any change in temperature.