Science Notes
Heat Notes 6th Science Lesson 7 Notes in English
6th Science Lesson 7 Notes in English
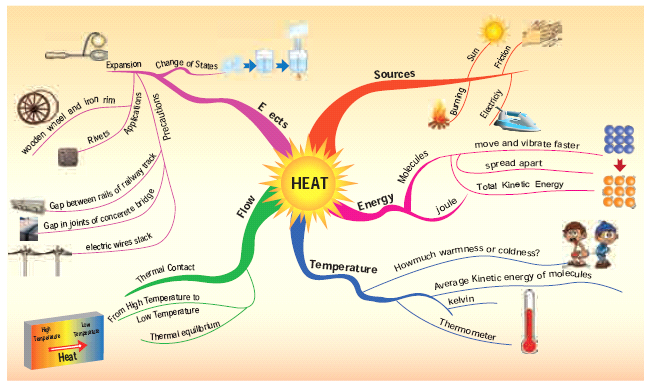
7] Heat
Introduction
- We are all familiar with heat. We feel it on our body when the sun shines, we use heat for cooking our food, and we reduce the heat by adding ice cubes while preparing fruit juice.
Sources of heat
Sun

- We all know that the sun gives us light. After standing under the sun light for some time, touch your head. It feels hot because the sun gives out heat besides light.
- Now, you can understand why it is difficult to walk bare-footed on sunny days in the afternoon.
Combustion (Burning)

- Heat energy can be generated by the burning of fuels like wood, kerosene, coal, charcoal, gasoline/petrol, oil, etc.,
Friction
- In the past people used to rub two stones together to light fire.
Electricity
- When electric current flows through a conductor, heat energy is produced. The water heater, iron box, electric kettle etc., work on this principle.

Heat
- Molecules in objects are constantly vibrating or moving inside objects.
- We cannot see that movement with our naked eye.
- When we heat the object this vibration and movement of molecules increases and temperature of the object also increases.

- Thus, Heat is an energy that raises the temperature of a thing by causing the molecules in that thing to move faster.
- Heat is not a matter. It doesn’t occupy space. It has no weight. Like light, sound and electricity, heat is a form of energy.
- In short, Heat is the total kinetic energy of constituent particles of objects. SI Unit of Heat is joule. The unit calorie is also used.
Hot and cold objects
- In our day-to-day life, we come across a number of objects. Some of them are hot and some of them are cold.
- How do we decide which object is hotter than the other? We use the tip of our finger to find out whether the tea in a cup has enough heat to drink or whether milk has been cooled enough to set for making curds. We often determine heat by touching the objects. But is our sense of touch reliable?
Temperature
Definition of Temperature
- The measurement of warmness or coldness of a substance is known as its Temperature.
- SI unit of temperature is Kelvin.
- Celsius and Fahrenheit are the other units used. Celsius is called as Centigrade as well.
- It determines the direction of flow of heat when two bodies are placed in contact.
Heat and Temperature
Heat and temperature is not the same thing, they in fact mean two different things;
- Temperature is related to how fast the atoms or molecules move or vibrate within the substance.
- Heat not only depends on the temperature of the substance but also depends on how many molecules are there in the object.
- Temperature measures the average kinetic energy of molecules. Heat measures the total Kinetic Energy of the molecules in the substance.
- Total heat is measured by calorie, the amount of heat needed to raise one gram of water by one degree centigrade.
Flow of Heat
An analogy between temperature and water level:
- Water ‘flows’ when there is a difference in the ‘levels’ of water in different places.
- It does not matter if there is more water in one place or another. Water from a puddle can flow into a reservoir or the other way around.
- The ‘temperature’ of an object is like the water level – it determines the direction in which ‘heat’ will flow.
- Heat energy flows from higher temperature to lower temperature.
Thermal contact and Thermal equilibrium
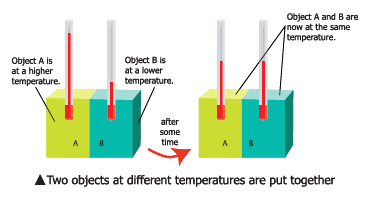
- Consider two bodies A and B. Let the temperature of A be higher than that of B.
- On bringing bodies A and B in contact, heat will flow from hot body A to the cold body B. Heat will continue to flow till both the bodies attain the same temperature.
- Two objects are said to be in thermal contact if they can exchange heat energy.
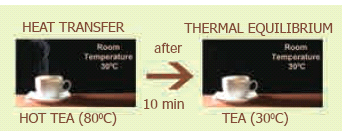
- Thermal equilibrium exists when two objects in thermal contact no longer affect each other’s temperature.
- For example, if a pot of milk from the refrigerator is set on the kitchen table, the two objects are in thermal contact. After certain period, their temperatures are the same, and they are said to be in thermal equilibrium.
Expansion in solids
- Most substances expand when heated and contract when cooled.
- The change in length / area or volume (due to contraction /expansion) is directly related to temperature change.
- The expansion of a substance on heating is called, the thermal expansion of that substance.
Linear and Cubical Expansion
- A solid has a definite shape, so when a solid is heated, it expands in all directions i.e., in length, area and volume, all increase on heating.
- The expansion in length is called linear expansion and the expansion in volume is called cubical expansion.
Uses of Thermal Expansion
Fitting the iron rim on the wooden wheel
- The diameter of the iron ring is slightly less than that of the wooden wheel.
- Therefore, it cannot be easily slipped on from the rim of wooden wheel.
- The iron ring is, therefore, first heated to a higher temperature so that it expands in size and the hot ring is then easily slipped over to the rim of the wooden wheel.
- Cold water is now poured on the iron ring so that it contracts in size and holds the wooden wheel tightly.

Riveting
- Rivets are used to join two steel plates together. Hot rivet is driven through the hole in the plates.
- One end of the rivet is hammered to form a new rivet head. When cooled, the rivet will contract and hold the two plates tightly together.

Cracking of a thick glass tumbler
- Glass is a poor conductor of heat.
- When hot liquid is poured into the tumbler, the inner surface of the tumbler becomes hot and expands while the outer surface remains at the room temperature and does not expand.
- Due to this unequal expansion, the tumbler cracks.
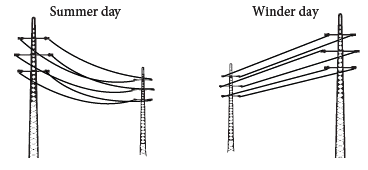
Electric wires
- Electric wires between electric posts contract on cold days and sag in summers.
- To solve this problem, we leave wires slack so that they are free to change length.
More to know:
- One day in 1922, the air temperature was measured at 59oC in the shade in Libya, Africa.
- The coldest temperature in the world was measured in the Antarctic continent. It was approximately – 89oC.
- The minus sign (–) is used when the temperature falls below the freezing point of water, which is 0oC.
- If water becomes ice at 0oC, you can imagine how cold – 89o C would be. Our normal body temperature is 37oC.
- Our body feels cool if the air temperature is around 15 to 20 degree Celsius.
- Glassware used in kitchen and laboratory are generally made up of Borosilicate glass (Pyrex glass).
- The reason is that the Borosilicate glasses do not expand much on being heated and therefore they do not crack.