Science Notes
ATOMIC STRUCTURE Notes 9th Science Lesson 8 Notes in English
ATOMIC STRUCTURE
Introduction
- Just as a small child wants to take a toy apart to find out what is inside, scientists have for long been curious about the internal structure of an atom.
- They wanted to find out what are the particles present inside an atom and how are these particles arranged in an atom.
- For explaining this many scientists proposed various atomic models.
- We have learnt Dalton’s atomic theory and J.J. Thomson’s model in class VIII.
- Now we will learn about sub-atomic particles and the other atomic models to explain how these particles are arranged within an atom.
Discovery of Nucleus
- In 1911, Lord Rutherford, a scientist from New Zealand, performed his famous experiment of bombarding a thin gold foil with very small positively charged particles called alpha (a) particles.
- He selected a gold foil because, he wanted as thin layer as possible and gold is the most malleable metal.
- He observed that:
- Most of the alpha particles passed straight through the foil.
- Some alpha particles were slightly deflected from their straight path.
- Very few alpha particles completely bounced back.
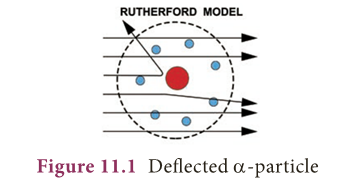

- Later, Rutherford generalized these results of alpha particles scattering experiment and suggested a model of the atom that is known as Rutherford’s Atomic model.
Rutherford’s Atomic model
- According to this model :
- The atom contains large empty space.
- There is a positively charged mass at the centre of the atom, known as nucleus.
- The size of the nucleus of an atom is very small compared to the size of an atom.
- The electrons revolve around the nucleus in close circular paths called orbits.
- An atom as a whole is electrically neutral, i.e., the number of protons and electrons in an atom are equal.
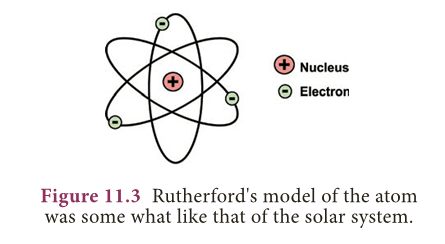
- Rutherford’s model of atomic structure is similar to the structure of the solar system.
- Just as in the solar system, the Sun is at the centre and the planets revolve around it, similarly in an atom the nucleus present at the centre and the electrons revolve around it in orbits or shells.
Limitations in Rutherford’s model
- According to Electromagnetic theory, a moving electron should accelerate and continuously lose energy.
- Due to the loss of energy, path of electron may reduce and finally the electron should fall into the nucleus.
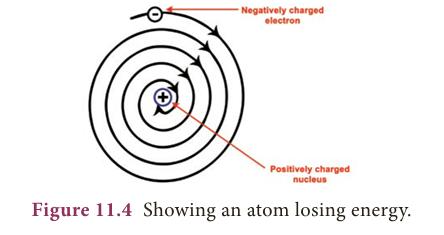
- If it happens so, atom becomes unstable. But atoms are stable.
- Thus, Rutherford’s model failed to explain the stability of an atom.
Bohr’s model of an atom
- In 1913, Neils Bohr, a Danish physicist, explained the causes of the stability of the atom in a different manner. The main postulates are:
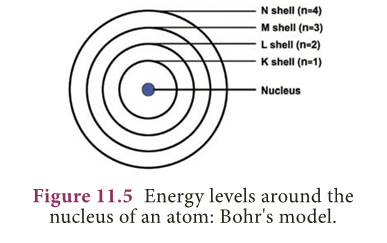
- In atoms, the electron revolve around the nucleus in stationary circular paths called orbits or shells or energy levels.
- While revolving around the nucleus in an orbit, an electron neither loses nor gains energy.
- An electron in a shell can move to a higher or lower energy shell by absorbing or releasing a fixed amount of energy.
- The orbits or shells are represented by the letters K,L,M,N,… or the numbers, n= 1,2,3,4,….
- The orbit closest to the nucleus is the K shell. It has the least amount of energy and the electrons present in it are called K electrons, and so on with the successive shells and their electrons.
- These orbits are associated with fixed amount of energy, so Bohr called them as energy level or energy shells.
Limitations of Bohr’s model
- One main limitation was that this model was applicable only to hydrogen and hydrogen like ions (example, He+, Li2+, Be3+, and so on). It could not be extended to multi electron nucleus.
Discovery of Neutrons
![]()
- In 1932 James Chadwick observed when Beryllium was exposed to alpha particles, particles with about the same mass as protons were emitted.
- These emitted particles carried no electrical charges.
- They were called as neutrons.
- The superscript 1 represents its mass and subscript 0 represents its electric charge.

Properties of Neutrons
- This particle was not found to be deflected by any magnetic or electric field, proving that it is electrically neutral.
- Its mass is equal to 1.676 × 10-24 g (1 amu).
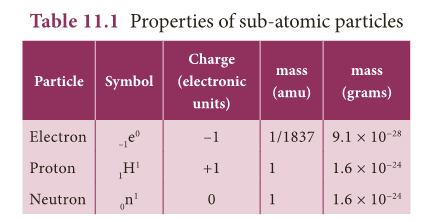
Characteristics of Fundamental particles
- The atom is built up of a number of sub- atomic particles.
- The three sub-atomic particles of great importance in understanding the structure of an atom are electrons, protons and neutrons, the properties of which are given in Table 11.1.
- There are two structural parts of an atom, the nucleus and the empty space in which there are imaginary paths called orbits.
- Nucleus: The protons and neutrons [collectively called nucleons] are found in the nucleus of an atom.
- Orbits: Orbit is defined as the path, by which electrons revolve around the nucleus.

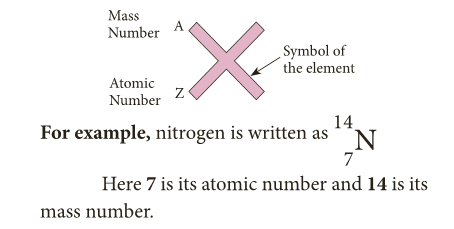
Atomic number and Mass number
- Only hydrogen atoms have one proton in their nuclei. Only helium atoms have two protons.
- Indeed, only gold atoms have 79 protons.
- This shows that the number of protons in the nucleus of an atom decides which element it is.
- This very important number is known as the atomic number (proton number, given the symbol Z) of an atom.
- Atomic number(Z) = Number of protons = Number of electrons
- Protons alone do not make up all of the mass of an atom.
- The neutrons in the nucleus also contribute to the total mass.
- The mass of the electron can be regarded as so small that it can be ignored.
- As a proton and a neutron have the same mass, the mass of a particular atom depends on the total number of protons and neutrons present. This number is called the mass number (or nucleon number, given the symbol A) of an atom.
- Mass number = Number of protons + Number of neutrons
- For any element, the atomic numbers are shown as subscripts and mass number are shown as superscripts.


Electronic configuration of atoms
- You already know that electrons occupy different energy levels called orbits or shells.
- The distribution of electrons in different shells is called electronic configuration.
- This distribution of electrons is governed by certain rules or conditions, known as Bohr and Bury Rules of electronic configuration.
- Rule 1: The maximum number of electrons that can be accommodated in a shell is equal to 2n2 where ‘n’ is the serial number of the shell from the nucleus.
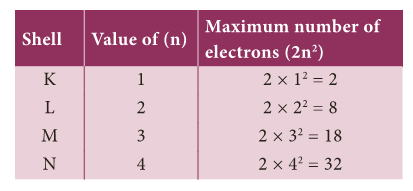
- Rule 2: Shells are filled in a stepwise manner in the increasing order of energy.
- Rule 3: The outermost shell of an atom cannot have more than 8 electrons , even if it has capacity to accomodate more electrons.
- For example, electronic arrangement in calcium having 20 electrons is,
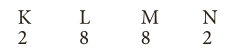

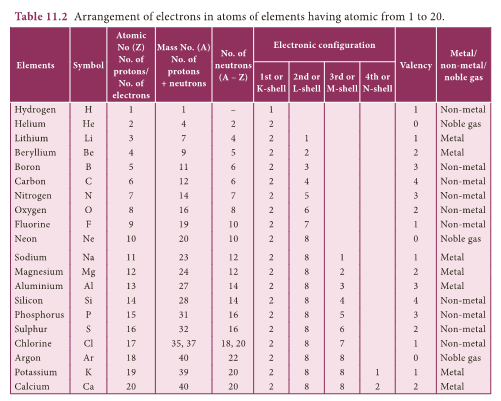
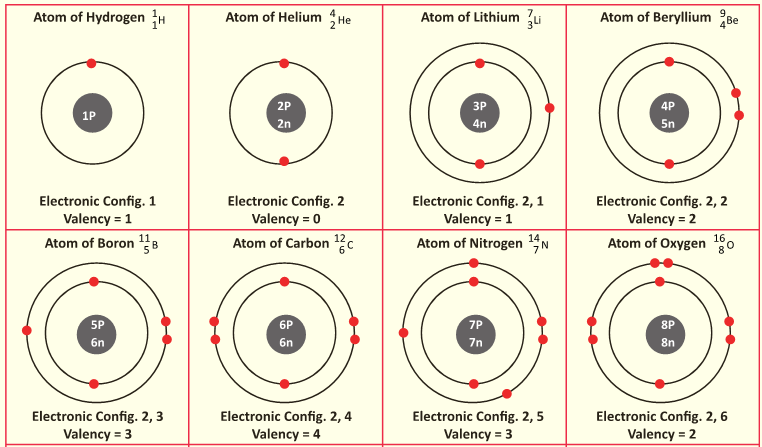
Geometric Representation of atomic structure of elements
- Knowing the mass number and atomic number of an element we can represent atomic structure.
- Valence electrons In the above example, we can see that there are six electrons in the outermost shell of oxygen atom.
- These six electrons are called as valence electrons.

- The outermost shell of an atom is called valence shell and the electrons present in the valence shell are known as valence electrons.
- The chemical properties of elements are decided by these valence electrons, since they are the ones that take part in chemical reactions.
- The elements with same number of electrons in the valence shell show similar properties and those with different number of valence electrons show different chemical properties.
- Elements, which have 1 or 2 or 3 valence electrons (except Hydrogen) are metals. Elements with 4 to 7 electrons in their valence shell are non-metals.
Valency
- Valency of an element is the combining capacity of the element with other elements and is equal to the number of electrons that take part in a chemical reaction.
- Valency of the elements having valence electrons 1, 2, 3, 4 is 1, 2, 3, 4 respectively.
- Valency of an element with 5, 6 and 7 valence electrons is 3, 2 and 1 (8–valence electrons) respectively.
- Because 8 is the number of electrons required by an element to attain stable electronic configuration.
- Elements having completely filled outermost shell show Zero valency.
- For example: The electronic configuration of Neon is 2,8 (completely filled).
- So valency is 0.

Isotopes, Isobars and Isotones
Isotopes

- In nature, a number of atoms of elements have been identified, which have the same atomic number but different mass numbers.
- For example, take the case of hydrogen atom, it has three atomic species as shown below:
- The atomic number of all the three isotopes is 1, but the mass number is 1, 2 and 3, respectively.

- On the basis of these examples, isotopes are defined as the different atoms of the same element, having same atomic number but different mass numbers.
- There are two types of isotopes: stable and unstable.
- The isotopes which are unstable, as a result of the extra neutrons in their nuclei are radioactive and are called radioisotopes.
- For example, uranium-235, which is a source of nuclear reactors, and cobalt-60, which is used in radiotherapy treatment are both radioisotopes.
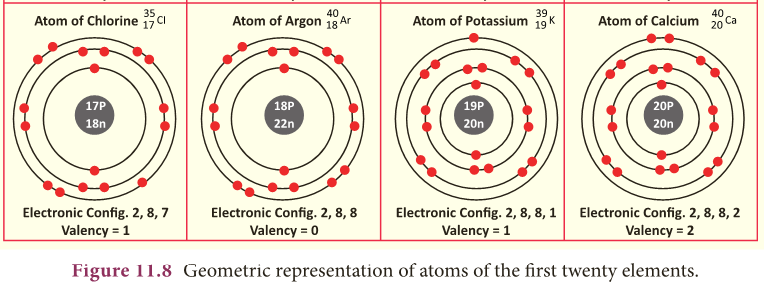
Isobars

- Let us consider two elements – calcium (atomic number 20), and argon (atomic number 18).
- They have (Fig. 11.10) different number of protons and electrons.
- But, the mass number of both these elements is 40.
- It follows that the total number of nucleons in both the atoms are the same.
- They are called isobars.
- Atoms of different elements with different atomic numbers, and same mass number are known as isobars.
Isotones
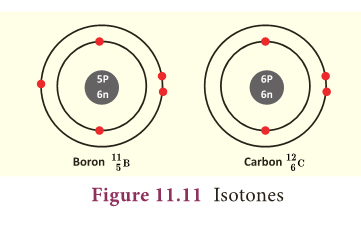
- No of neutrons in boron = 11 – 5 = 6
- No of neutrons in carbon = 12 – 6 = 6
- The above pair of elements Boron and Carbon has the same number of neutrons but different number of protons and hence different atomic numbers.
- Atoms of different elements with different atomic numbers and different mass numbers, but with same number of neutrons are called isotones
Laws of Chemical combination
- In the seventeenth century, scientists had been trying to find out methods for converting one substance into another.
- During their studies of chemical changes, they made certain generalisations.
- These generalisations are known as laws of chemical combination.
- These are :
- Law of conservation of mass
- Law of constant proportions
- Law of multiple proportions
- Law of reciprocal proportions
- Gay Lussac’s law of gaseous volumes
- Out of these five laws you have already learnt the first two laws in class VIII.
- Let us see the next three laws in detail in this chapter.
Law of multiple proportions
- This law was proposed by John Dalton in 1804. It states that, “When two elements A and B combine together to form more than one compound, then different masses of A which separately combine with a fixed mass of B are in simple ratio”.
- To illustrate the law let us consider the following example.
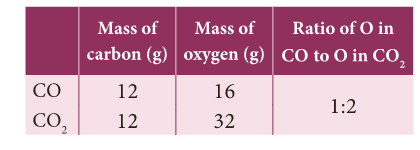
- Carbon combines with oxygen to form two different oxides, carbon monoxide(CO) and carbon dioxide (CO2).
- The ratio of masses of oxygen in CO and CO2 for fixed mass of carbon is 1: 2.
- Let us take one more example, Sulphur combines with oxygen to form sulphur dioxide and sulphur trioxide.
- The ratio of masses of oxygen in SO2 and SO3 for fixed mass of Sulphur is 2:3.
Law of Reciprocal Proportions
- The law of reciprocal proportions was proposed by Jeremias Ritcher in 1792.
- It states that, “If two different elements combine separately with the same weight of a third element, the ratio of the masses in which they do so are either same or a simple multiple of the mass ratio in which they combine among themselves.”
- Consider the three elements hydrogen, oxygen and water as shown below:
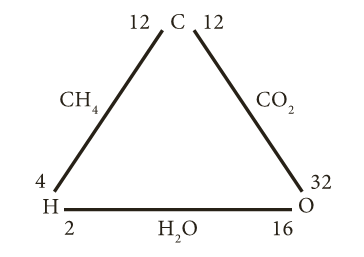
- Here, hydrogen and oxygen combine separately with the same weight of carbon to form methane (CH4) and carbon dioxide (CO2)

- Now, hydrogen and oxygen combine to form water (H2O).
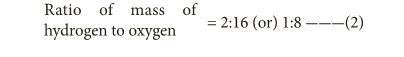
- From 1 and 2, the ratio is the same as that of the first obtained. Thus, the law of reciprocal proportion is illustrated.
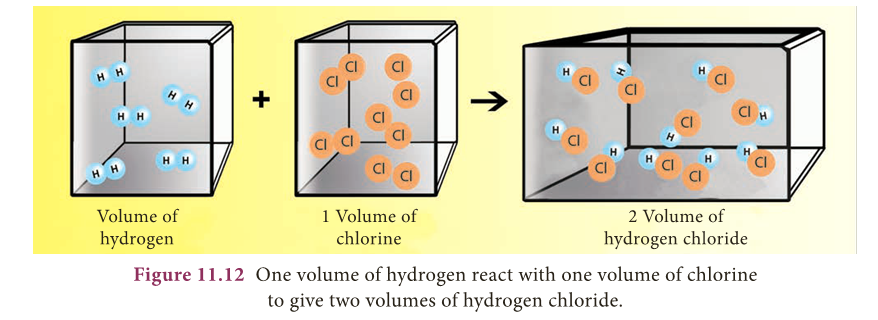
Gay Lussac’s Law of Combining Volumes
- According to Gay Lussac’s Law, whenever gases react together, the volumes of the reacting gases bear a simple ratio, and the ratio is extended to the product when the product is also in gaseous state, provided all the volumes are measured under similar conditions of temperature and pressure.
- This law may be illustrated by the following example.
- It has been experimentally observed that two volumes of hydrogen reacts with one volume of oxygen to form two volumes of water as shown in the figure 11.12.
- The ratio of volume which gases bears is 2:1:2 which is a simple whole number ratio.
- Quantum Numbers
- When you specify the location of a building, you usually list which country it is in, which state and city it is in that country.
- Just like we have four ways of defining the location of a building (country, state, city, and street address), we have four ways of defining the properties of an electron, i.e.four quantum numbers.
- Thus, the numbers which designate and distinguish various atomic orbitals and electrons present in an atom are called quantum numbers.
- Four types of Quantum number are as follows:
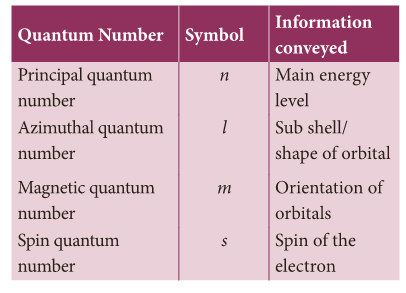
- You will learn more details about this in higher classes.
MORE TO KNOW:
The nucleus as neutral.
- In 1920 Rutherford predicted the presence of another particle in the nucleus as neutral.
- James Chadwick, the inventor of neutron was student of Rutherford
Atomic number is designated as Z why?
- Z stands for Zahl, which means NUMBER in German.
- Z can be called Atomzahl or atomic number A is the symbol recommened in the ACS style guide instead of M (massenzahl in German).
The Nucleus
- Besides the fundamental particles like protons, electrons and neutrons some more particles are discovered in the nucleus of an atom.
- They include mesons, neutrino, antineutrino, positrons etc.
AMU
- Atoms are so tiny their mass number cannot be expressed in grams but expressed in amu (atomic mass unit). New unit is U.
- Size of an atom can be measured in nano metre (1 nm = 10-9 m)
- Even though atom is an invisible tiny particle now-a-days atoms can be viewed through SEM that is Scanning Electron Microscope.
Yukawa forces
- The forces between the protons and the neutrons in the nucleus are of special kind called Yukawa forces.
- This strong force is more powerful than gravity.
Thumb rule
- Thumb rule for isotopes and isobars. Remember t for top and b for bottom.
- Isotope: Top value changes – Atomic mass Isobars: Bottom value changes – Atomic number
EXTRA POINTS:
- Atom: The smallest component of an element which takes part in a chemical reaction.
- Electron: A stable subatomic particle with a charge of negative electricity, found in all atoms and acting as the primary carrier of electricity in solids.
- Neutron: A subatomic particle of about the same mass as a proton but without an electric charge, present in all atomic nuclei except those of ordinary hydrogen.
- Orbitals: Atomic orbitals are region of space around the nucleus of an atom where an electron is likely to be found.
- Proton: A stable subatomic particle occurring in all atomic nuclei, with a positive electric charge equal in magnitude to that of an electron.
- Quantum numbers: The numbers which designate and distinguish various atomic orbitals and electrons present in an atom.