7th Std Science Lesson Wise Questions in English – Part 1
7th Science Lesson 4 Questions in English
4] Atomic Structure
1. What is an average diameter of atom?
- 1×10-2 m
- 1×10-4 m
- 1×10-7 m
- 1×10-10 m
Explanation
An atom is one and thousand times smaller than the thickest human hair. It has an average diameter of 0.0000000001m or 1×10-9 m.
2. Who proposed the atomic theory in the year 1808?
- Rutherford
- Chadwick
- John Dalton
- J. J. Thomson
Explanation
John Dalton proposed the atomic theory in the year 1808. He proposed that matter consists of very small particles which he named atoms.
3. One nanometre is equal to __________
- 1×10-9 m
- 1×10-10 m
- 1×109 m
- 1×1010 m
Explanation
Nanometre is the smallest unit used to measure small lengths. One metre is equal to 1×109 nm or one nanometre is equal to 1×10-9 m.
4. John Dalton proposed atom is smallest indivisible particle and in what shape?
- Cylindrical
- Square
- Spherical
- Trapezium
Explanation
John Dalton proposed an atom is smallest indivisible particle, it is spherical in shape. His theory does not propose anything about the positive and negative charges of an atom.
5. Who proposed different atomic theory in 1897 and compared an atom to a watermelon?
- Rutherford
- Chadwick
- J. J. Thomson
- Goldstein
Explanation
In 1897 J.J Thomson proposed a different theory. He compared an atom to a watermelon.
6. Which among the following statement is correct
- J. J. Thomson theory proposed that the atom has negatively charged part like the red part of the watermelon and in it are embedded, like the seeds, positively charged particles which he called protons.
- According to this theory as the positive and negative charges are equal, the atom as a whole does not have any resultant charge. Thomson’s greatest contribution was to prove by experimentation the existence of the negatively charged particles or electrons in an atom.
- Only 1
- Only 2
- Both 1 and 2
- None
Explanation
J. J. Thomson theory proposed that the atom has positively charged part like the red part of the watermelon and in it are embedded, like the seeds, negatively charged particles which he called electrons.
7. Who conducted an experiment bombarded a very thin layer of gold with positively charged alpha rays?
- J. J. Thomson
- Goldstein
- James Chadwick
- Rutherford
Explanation
There were short coming in Thomson’s theory, Earnest Rutherford gave a better understanding. Earnest Rutherford conducted an experiment. He bombarded a very thin layer of gold with positively charged alpha rays.
8. Which among the following statement is correct
- Rutherford bombarded a very thin layer of gold with positively charged alpha rays. He found that most of these rays which travel at a great velocity passed through the gold sheet without encountering any obstacles. A few are, however, turned back from the sheet.
- Based on this experiment, Rutherford proposed his famous theory. In his opinion, – The fact that most alpha particles pass through the gold sheet means that the atom consists mainly of electron in it.
- The part from which the positively charged particles are turned back is positively charged but very small in size as compared to the empty space.
- Both 1 and 2
- Both 1 and 3
- Both 2 and 3
- All 1, 2 and 3
Explanation
Based on this experiment, Rutherford proposed his famous theory. In his opinion, – 1. The fact that most alpha particles pass through the gold sheet means that the atom consists mainly of empty space.
9. Which among the following is not the theory proposed by Rutherford
- The nucleus at the centre of the atom has the positive charge.
- Most of the mass of the atom is concentrated in the electrons.
- The negatively charged electrons revolve around the nucleus in specific orbits.
- In comparison with the size of the atom, the nucleus is very, very small.
Explanation
Rutherford’s theory proposes that 1. The nucleus at the centre of the atom has the positive charge. Most of the mass of the atom is concentrated in the nucleus. 2. The negatively charged electrons revolve around the nucleus in specific orbits. 3. In comparison with the size of the atom, the nucleus is very, very small.
10. Which among the following is not the Subatomic Particle?
- Proton
- Electron
- Nucleus
- Neutron
Explanation
The discoveries made during the twentieth century proved that atoms of all elements are made up of smaller components – electron, proton and neutron. An electron from hydrogen atom is no different from electron of a carbon atom. In the same manner, protons and neutrons of all elements also have same characteristics. These, particles that make up the atom are called Subatomic Particles.
11. Which among the following particle is present in nucleus?
- Only proton
- Proton and Electron
- Only Electron
- Proton and Neutron
Explanation
The proton is the positively charged particle and its located in the nucleus. Its positive charge is of the same magnitude as that of the electron’s negative charge. Neutron is inside the nucleus.
12. Which revolve around the nucleus of the atom in specific orbits?
- Proton
- Electron
- Positron and proton
- Both proton and neutron
Explanation
Electron is a negatively charged particle. Electrons revolve around the nucleus of the atom in specific orbits.
13. The nuclei of all atoms contain neutrons except which one?
- Hydrogen
- Oxygen
- Nitrogen
- Helium
Explanation
Neutron is inside the nucleus. The neutron does not have any charge. Excepting hydrogen (protium), the nuclei of all atoms contain neutrons.
14. Protons and Neutrons are the two types of particles in the nucleus of an atom. They are called ___________
- Positrons
- Nucleons
- Magnetron
- None of the above
Explanation
Protons and Neutrons are the two types of particles in the nucleus of an atom. They are called nucleons.
15. Which among the following statement is correct
- The total negative charge of all an electron outside the nucleus is less than the total positive charge in the nucleus.
- The total negative charge of all an electron outside the nucleus is equal to the total positive charge in the nucleus.
- The total negative charge of all an electron outside the nucleus is more than total positive charge in the nucleus.
- The total negative charge of all an electron outside the nucleus where there is only one positive charge in the nucleus.
Explanation
The total negative charge of all an electron outside the nucleus is equal to the total positive charge in the nucleus. That makes the atom electrically neutral.
16. Which among the following particle is not correctly matched with its scientist who discovered?
- Proton – Dalton
- Electron – J. J. Thomson
- Neutron – Rutherford
- Only 1
- Both 1 and 2
- Both 1 and 3
- All 1, 2 and 3
Explanation
Proton(p) – Ernest Rutherford
Electron(e) – Sir John Joseph Thomson
Neutron(n) – James Chadwick
17. What is the mass of proton?
- 1.5298 ×10-27 kg
- 1.6726 ×10-27 kg
- 4.8173 ×10-27 Kg
- 9.1093 × 10-27 kg
Explanation
The mass od proton is 1.6726 ×10-27. Its charge is +1.
18. What is the mass of Electron?
- 1.6749 ×10-31 kg
- 3. 761 ×10-31 kg
- 4.8173 ×10-31 Kg
- 9.1093 × 10-31 kg
Explanation
The mass of Electron is 9.1093 × 10-31. The charge of electron is –1.
19. The number of electrons or protons in an atom is called _________
- Mass number
- Atomic number
- Nuclei number
- All the above
Explanation
The number of electrons or protons in an atom is called the atomic number of that atom.
20. What is the mass of neutron?
- 1.6749 ×10-27 kg
- 3. 761 ×10-27 kg
- 4.8173 ×10-27 Kg
- 9.1093 × 10-27 kg
Explanation
The mass of Neutron is 1.6749 × 10-27 kg. The charge of neutron is 0 i.e., no charge.
21. The Atomic number is represented by which letter?
- A
- Z
- T
- M
Explanation
The atomic number is represented by the letter Z. if we know the atomic number of an atom, we know the number of electrons or protons in it.
22. What is the atomic number of Oxygen?
- 4
- 6
- 8
- 12
Explanation
In the Oxygen atom there are eight protons and eight electrons in orbit around the nucleus, so the atomic number of helium is z=8.
23. Which is equal to the sum of the number of protons(p) and neutrons (n) in the nucleus?
- Mass number
- Atomic number
- Nuclei number
- All the above
Explanation
Mass number is equal to the sum of the number of protons(p) and neutrons (n) in the nucleus.
Atomic mass or mass number = Number of Protons + Number of Neutrons
24. The mass of an atom is concentrated in what?
- Neutron
- Positron
- Nucleus
- Electron
Explanation
The mass of an atom is concentrated in its nucleus.
25. The mass number is represented by which letter?
- A
- Z
- M
- T
Explanation
The Mass number is represented by letter A.
26. What is the atomic mass number of Lithium (3 Protons and 4 neutrons)?
- 1
- 7
- 9
- 12
Explanation
A lithium atom contains 3 Protons and 4 neutrons. Its atomic mass number A = 3+4 = 7.
27. Atoms that have the same mass number but different atomic numbers is known as ____
- Isotopes
- Isobars
- Isometrics
- Isotonic
Explanation
Atoms that have the same mass number but different atomic numbers is known as Isobars. for example, Calcium – 40 and Argon – 40.
28. In a sodium atom, there are 11 Protons and 12 neutrons. What is the atomic mass?
- 11
- 12
- 23
- 1
Explanation
In a sodium atom, there are 11 Protons and 12 neutrons. Hence, its atomic mass number A = 11 + 12 = 23.
29. Atoms that have same atomic number but different mass numbers is known as _____
- Isotopes
- Isobars
- Isometrics
- Isotonic
Explanation
Atoms of the same element can have different number of neutrons. Such atoms will have same atomic number but different mass numbers. These atoms are called isotopes. For example Hydrogen has three isotopes — Hydrogen (1H1), Deuterium (1H2), Tritium (1H3).
30. In Nitrogen (7N14) which is the atomic number?
- 7
- 14
- 21
- None of the above
Explanation
7 is the atomic number and 14 is the mass number.
31. Which among the following element is not correctly matched with its mass number?
- Hydrogen – 1
- Helium – 4
- Aluminium – 12
- Oxygen – 16
Explanation
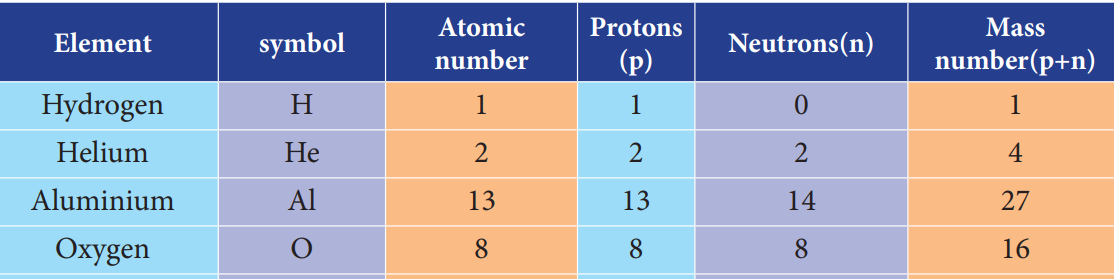
32. If the atomic number of sodium is 11 and mass number is 23, then what is the number electron in sodium atom?
- 11
- 12
- 34
- 5
Explanation
In an atom no of proton and no of electrons are same i.e., sodium has 11 proton and 11 electrons. So, its atomic number is 11.
33. Some atoms can hold one electron, some can hold two, some can hold three, some can hold four and some cannot hold any electron. This property is called ___________
- Isotone
- Fusion
- Barrer
- Valency
Explanation
Some atoms can hold one electron, some can hold two, some can hold three, some can hold four and some cannot hold any electron. This property is called valency.
34. Valency of an atom is measured by its combining property with which among the following atoms?
- Helium
- Hydrogen
- Lithium
- Oxygen
Explanation
The combining property of an atom is called as Valency. It is a measure of how many hydrogen atoms it can combine with.
35. What is the valency of Oxygen?
- One
- Two
- Three
- Four
Explanation
Oxygen can combine with two hydrogen atoms and create water molecule; the valency of oxygen atom is two.
36. In chlorine, it combines with how many hydrogen atoms to create Hydrochloric acid?
- One
- Two
- Three
- Four
Explanation
In case of chlorine, it can combine with only one hydrogen to create HCl (hydrochloric acid) here the valency of chlorine is one.
37. Methane has one carbon atom combining with how many hydrogen atoms?
- One
- Two
- Three
- Four
Explanation
Methane has one carbon atom combining with four hydrogen atoms to form carbon molecule is methane (CH4).
38. Which among the following statement is correct
- Valency is defined as the combining capacity of an element. Atoms of different elements combine with each other to form molecules. Valency determines the number of atoms of an element that combines with atom or atoms of another type.
- Electrons carry a negative electric charge, and protons carry a positive charge. The attraction between them holds electrons in orbits.
- Only 1
- Only 2
- Both 1 and 2
- None
39. Which among the following is the Monovalent?
- Sodium
- Oxygen
- Beryllium
- Calcium
Explanation
The element having valency one is called monovalent. For example: Hydrogen and Sodium.
40. Which among the following element is present in Banana?
- Carbon
- Helium
- Potassium
- Argon
Explanation
Potassium is the element present in Banana. Its atomic number is 19.
41. Which among the following is not the divalent?
- Nitrogen
- Beryllium
- Oxygen
- Calcium
Explanation
The elements having valency two are called divalent. For example: Oxygen, calcium and Beryllium.
42. Which among the following is used to fill the balloons that float?
- Lithium
- Helium
- Neon
- Fluorine
Explanation
Helium is used to fill the balloon that floats.
43. Which among the following is used in crackers and match sticks?
- Neon
- Fluorine
- Carbon
- Phosphorous
Explanation
Phosphorus is used in crackers and match sticks.
44. Which among the following statement is incorrect
- When atoms of different elements combine with each other than molecules of compounds are formed. In these instances, it is necessary to know the valency of those elements. For example: 2 Na + Cl2 ———- 2NaCl
Valency 1 + 1
- Here, the valency’s of both sodium and chlorine are 1. Remember The valency of element Na is 1 The valency of element Cl is 1 Then, the molecular formula will be Symbol of Elements NaCl Molecular Formula; Radicals and ions = 1 and 1.
- Only 1
- Only 2
- Both 1 and 2
- None
45. Which among the following is the trivalent?
- Aluminium
- Carbon
- Hydrogen
- Calcium
Explanation
The elements having valency three are called trivalent. For example: Nitrogen and Aluminium. Some elements exhibit more than one valency. For example: Iron combines with oxygen to form two types ferrous oxide (exhibits valency 2) and ferric oxide (exhibits valency 3).
46. In Mg + Cl2 —????- Mg Cl2. What is the valency of Mg?
- 1
- 2
- 4
- 3
Explanation
Here, the valency of magnesium is 2 and that of Cl is 1.